PeekMed, a Braga startup working in the medical field, receives approval from the Food and Drug Administration (FDA) to introduce its 3D orthopedic surgery planning system into the US market.
“PeekMed was designed with surgeons for surgeons”
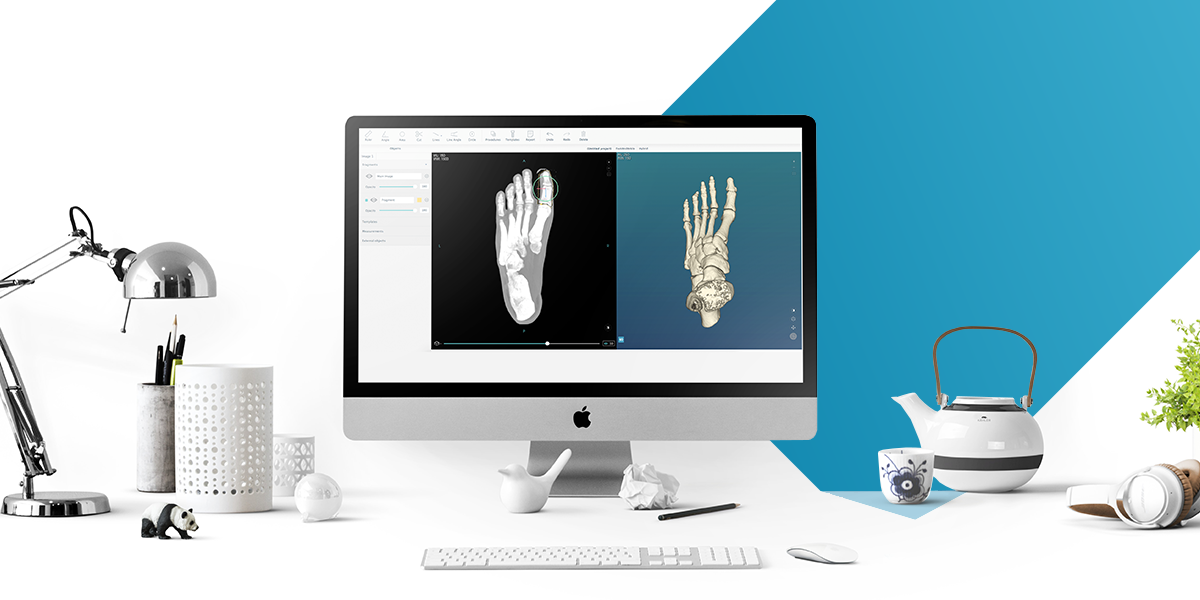
PeekMed provides 3D orthopedic surgical planning tools. This technology helps the orthopedic surgeon to intuitively and freely manipulate the 3D model created through the imaging study of the patient.
The osteosynthesis material of various companies can be added to the planning in the system, allowing the orthopedic surgeon to analyze the surgery’s impact.
Read More: PeekMed launches first 3D Orthopedic Software in the world
With the FDA certification, health professionals all across the US will be able to digitally perform the surgical planning without changing their surgical process or requirements.

João Pedro Ribeiro
“PeekMed was designed with surgeons for surgeons. Therefore, this is an incredible achievement for the company as it allows us to deliver the product to the largest market globally!” exclaimed PeekMed CEO João Pedro Ribeiro, in a statement.
“Our goal is to provide the best pre-operative planning system to improve the outcomes of the 6.6 million surgeries expected in 2020. This now continues with every single surgeon across the US,” he added.
Only a few European companies achieve certification in the US due to the detailed evaluation and responsibility attributed by this certification.
The Portuguese startup’s system uses any standard patients’ medical images, such as X-Rays, CT or MRI scan, which can be imported from a CD ROM, a local folder or local PACS (Picture Archiving and Communication Systems).
“PeekMed is the first user-friendly system which enables precise surgical 3D planning”

François Lintz
“PeekMed is the first user-friendly system which enables precise surgical 3D planning. Being able to simulate surgical cuts and virtually adapt the implants to the patient’s own anatomy is really a game changer,” said Dr. François Lintz, Orthopedic surgeon and Chair in scientific committee at the French Foot and Ankle Society.
PeekMed reached 510(k) clearance for its orthopedic planning system, from the U.S. Food and Drug Administration, after regulatory approval in the European Union, and successfully working with more than 1,100 surgeons.
Accelerated at Startup Braga, PeekMed is a software solution developed for orthopedic surgeons that aims at optimizing the pre-operative planning process. By simulating the expected results prior to the surgery occurring, it allows for more accuracy in the actual surgery.
Read More: Startup Braga’s 115 startups have raised €26.8M to date
In 2015, the Portuguese startup was selected for investment by Portugal Ventures.
Read More: Portugal Ventures announced investment in Peekmed
Portugal Ventures is a venture capital firm that invests in seed rounds of Portuguese startups in tech, life sciences, and tourism.
A word from our Sponsor: Looking for Content Marketing support? Click here.